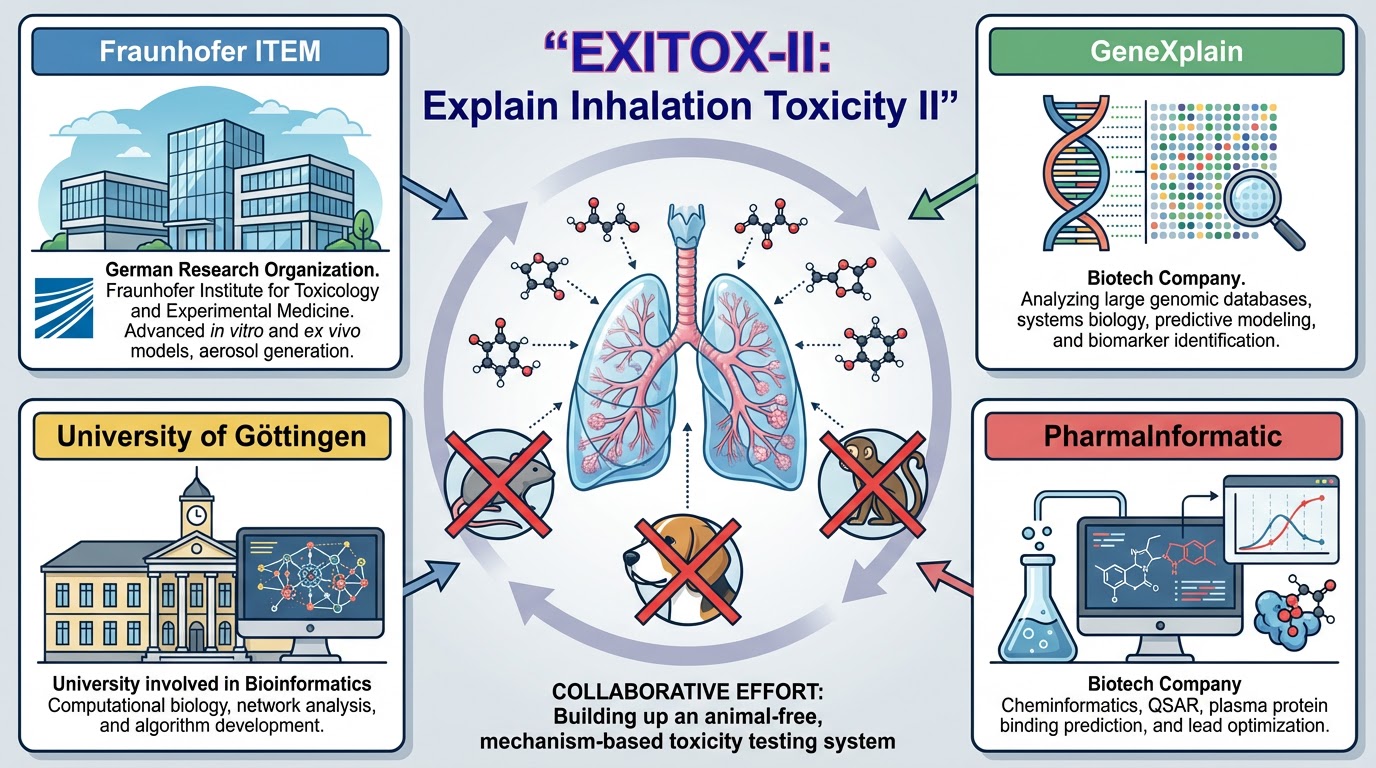
Animal-free mechanism-based toxicity testing – predict toxicity after repeated dose inhalation exposure by using a read across approach.
The collaboration between the University of Göttingen, geneXplain GmbH, PharmaInformatic, and the Fraunhofer Institute for Toxicology and Experimental Medicine (Fraunhofer ITEM) has developed an integrated approach for testing and assessment (IATA) aimed at replacing animal studies involving repeated inhalational exposure.
PharmaInformatic leveraged its platform technology to develop an expert system that evaluates pharmacokinetic and physiochemical properties of substances, such as plasma protein binding. Understanding the extent to which drugs and toxic substances bind to proteins in plasma or blood is crucial, as this binding reduces the free (effective) concentration of compounds in vivo. For toxicity evaluations, it is vital to know the degree of plasma protein binding for a compound.
The collaborative project was funded by the German Federal Ministry of Education and Research (BMBF) in the funding program “e:ToP – Innovative Toxikologie zur Reduzierung von Tierversuchen”.


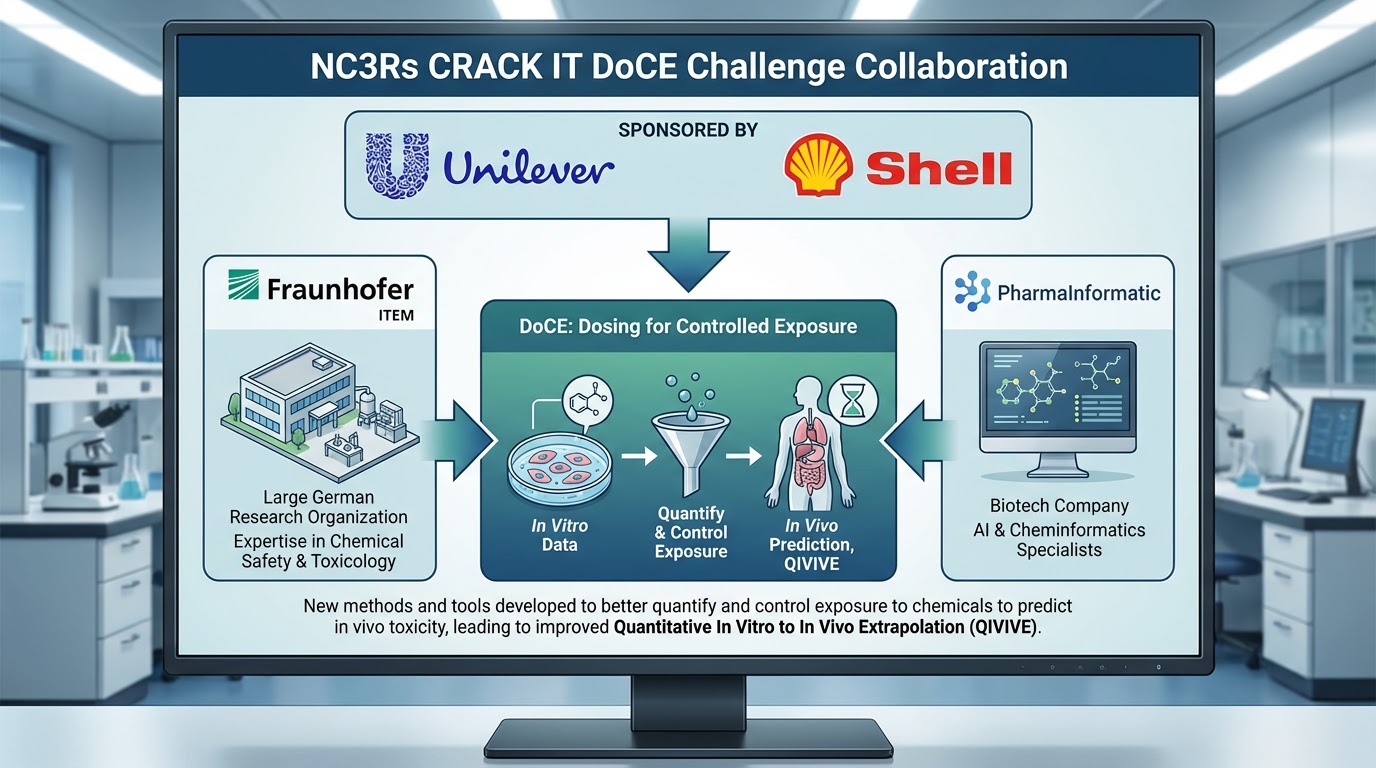
DoCE (Dosing for Controlled Exposure): This project focused on developing dosing strategies to characterize in vitro dose-responses with enhanced relevance for in vivo extrapolation. Sponsored by Unilever and Shell, the initiative successfully analyzed methods and tools to improve the quantification and control of chemical exposure for more accurate in vivo toxicity predictions.
The project achieved a better understanding of the relationship between in vitro concentrations and in vivo exposure, increasing the success of using in vitro approaches early in development.
As part of the CRACK IT Challenge, this project was supported by the NC3Rs (National Centre for the Replacement, Refinement and Reduction of Animals in Research). The NC3Rs is a UK-based organization dedicated to advancing biosciences by promoting alternatives to animal use in research and testing.



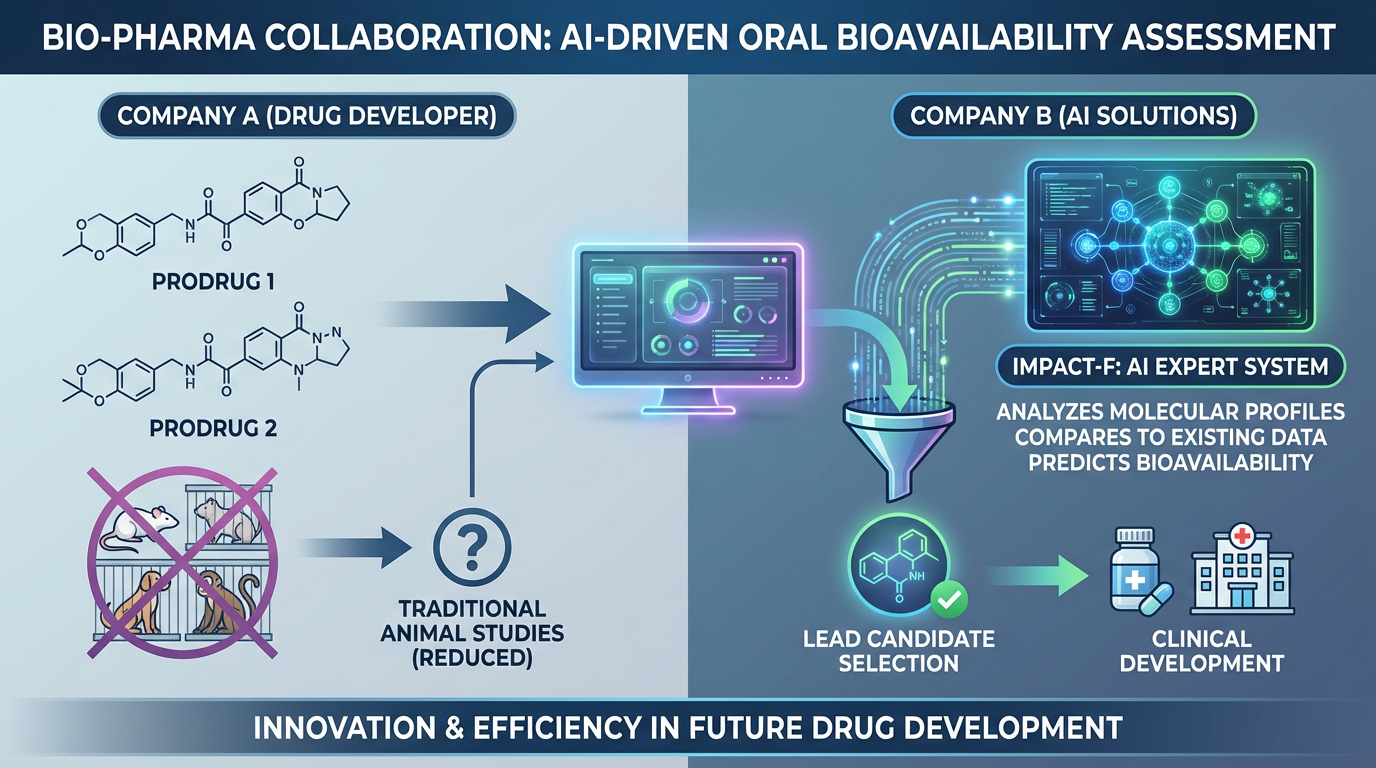
Avivia BV and PharmaInformatic have entered into a research collaboration agreement focused on optimizing cancer treatment options. Under this agreement, PharmaInformatic used its IMPACT-F system to estimate the human oral bioavailability of prodrugs in development.
Avivia BV required an efficient solution to evaluate the oral bioavailability of two different prodrugs for the same active drug, without relying heavily on traditional animal studies in rats, dogs, and monkeys. The IMPACT-F system analyzed the molecular profiles of our prodrugs, comparing them to existing data on chemically similar prodrugs and drugs. This insightful analysis enabled us to make an informed selection of the lead candidate, which has now progressed to clinical development.
Hans Platteeuw, CEO of Avivia BV, shared his positive experience, stating, “We were quite impressed with the IMPACT-F system; it truly helped us to better understand our prodrugs without extensive animal testing.”
This collaboration exemplifies the innovative power and efficiency of leveraging advanced computational models in drug development.
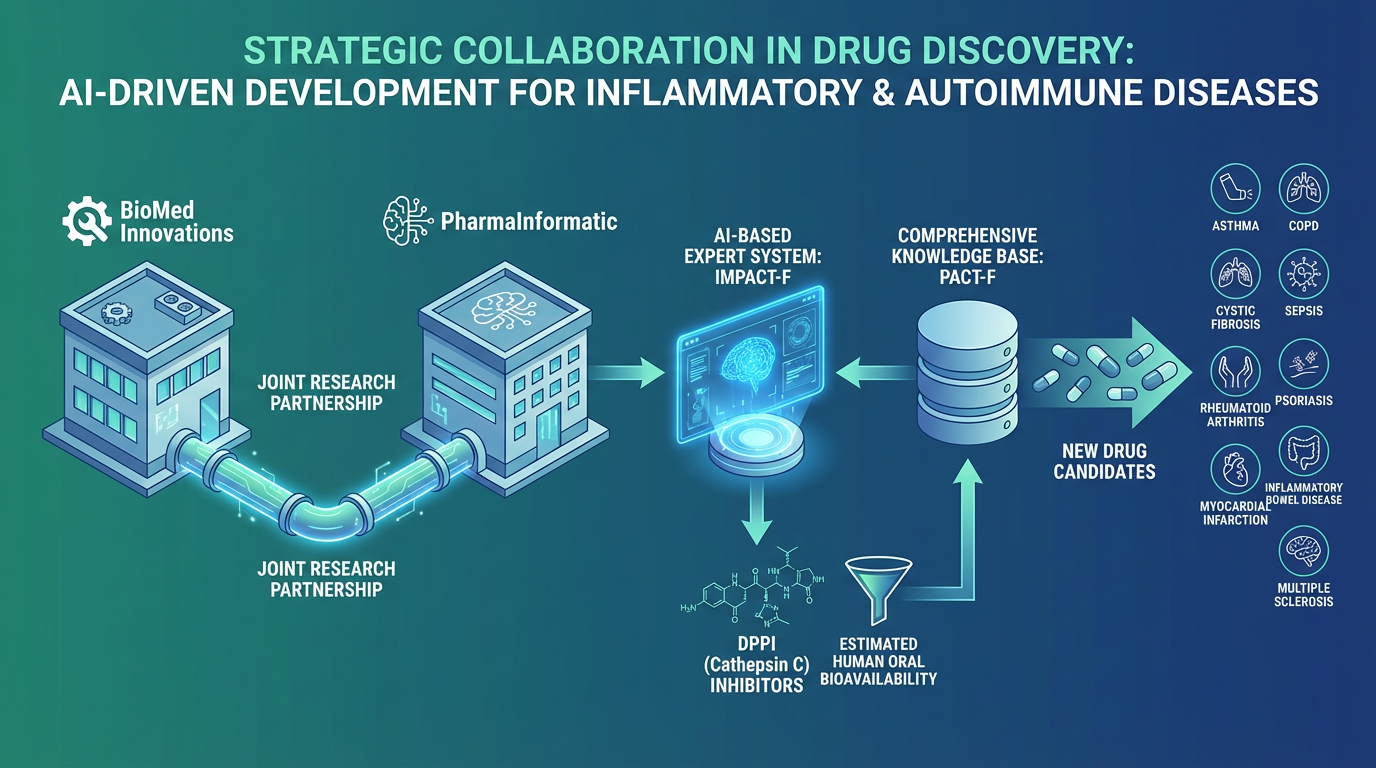
PharmaInformatic and UNIZYME Laboratories A/S have embarked on a research collaboration to advance the development of novel therapeutic agents targeting a range of inflammatory diseases.
Through this partnership, PharmaInformatic employs its expert system, IMPACT-F, to prioritize dipeptidyl peptidase I (DPPI, cathepsin C) inhibitors based on their estimated oral bioavailability in humans. DPPI, a significant therapeutic target, plays a crucial in the development of treatments for various inflammatory and autoimmune diseases.
Cathepsin C inhibitors show promise as potential therapeutics for conditions such as asthma, chronic obstructive pulmonary disease (COPD), cystic fibrosis, sepsis, rheumatoid arthritis, psoriasis, myocardial infarction, inflammatory bowel disease, and multiple sclerosis.
This research collaboration utilizes advanced AI methods, enriched by the comprehensive data and insights from the PACT-F knowledge base, to develop new drugs for addressing urgent medical needs.

PharmaInformatic has entered into a collaboration agreement with Bridge Bioresearch PLC, aimed at advancing drug development for the treatment of type 2 diabetes. Under this partnership, PharmaInformatic utilizes its expert system, IMPACT-F, to predict the oral bioavailability of promising drug candidates.
Søren Stenderup, CEO of Bridge Bioresearch, elaborates on this collaboration in BioPeople’s newsletter, Denmark:
“We are developing molecules for the treatment of Type 2 diabetes and had all necessary documentation except for human bioavailability data. Upon being approached by the German biotech company PharmaInformatic, which developed the expert system IMPACT-F for predicting oral bioavailability in humans, we saw a great opportunity. We collaborated with PharmaInformatic, and in a short time, our development project advanced to the next stage.”
Type 2 diabetes is a prevalent metabolic disorder affecting a significant portion of the global population. According to recent estimates, hundreds of millions of people worldwide are affected by diabetes, with approximately 90% suffering from type 2 diabetes.