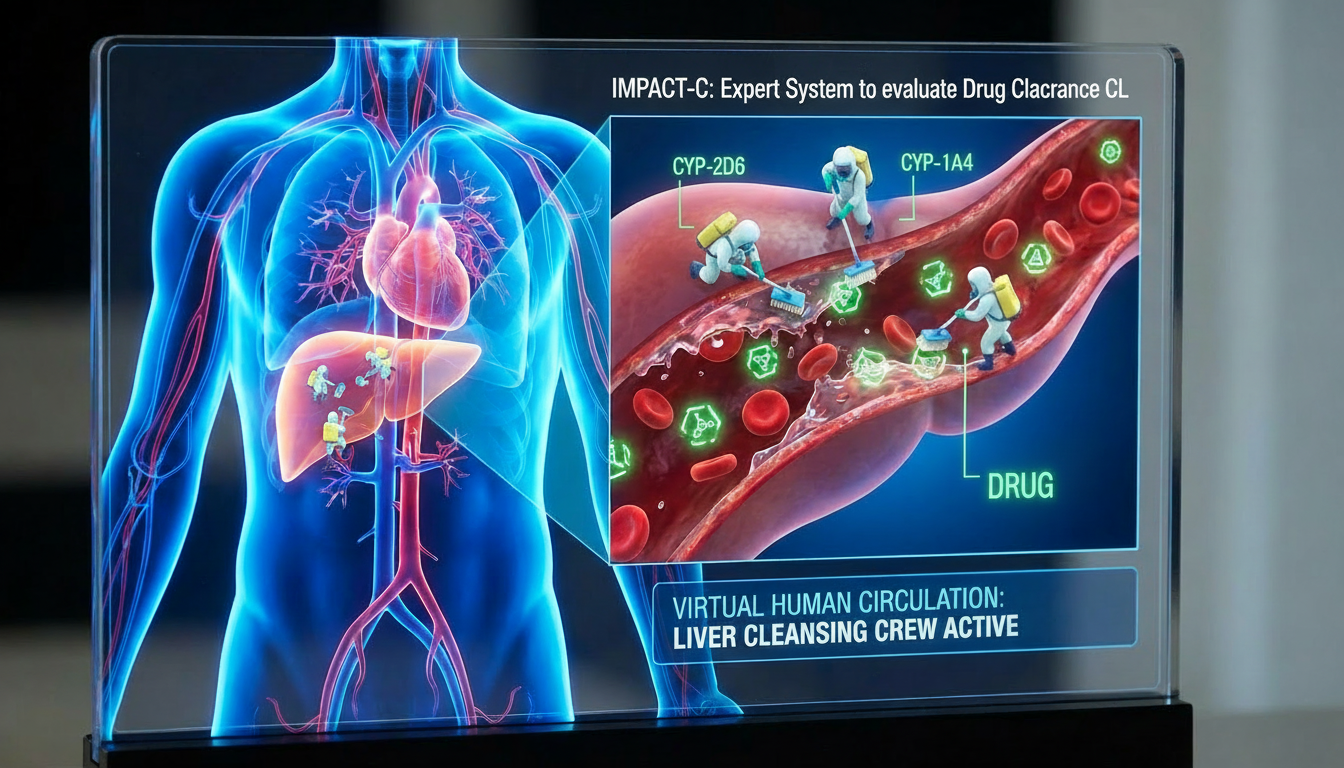
The expert system IMPACT-C assesses the clearance CL of a small-molecule drug candidate in humans and animals, including monkeys, dogs, and rats. This important pharmacokinetic parameter describes the efficiency of a drug’s elimination from the body.
Evaluating drug clearance offers several advantages:
Discover the accuracy of our predictions by validating the expert system with your own preclinical drug results in monkeys, dogs, and rats. It’s easy, confidential, and delivers reliable results quickly. Get in touch via our [contact form].
This AI-based expert system is grounded in the world’s largest knowledge base on drug clearance, known as PACT-C (Preclinical And Clinical Trials Knowledge Base on Clearance).
Clearance can affect the oral bioavailability of a drug (see IMPACT-F) due to the first-pass effect. This depends on the specific drug structure and the extent to which functional groups within the molecule are metabolized, primarily in the liver.

The expert system IMPACT-D predicts the Volume of Distribution (Vd) of small-molecule drug candidates in humans and animals, including monkeys, dogs, and rats. This crucial pharmacokinetic parameter describes how extensively a drug disperses throughout the body’s tissues relative to its concentration in the systemic circulation.
A high volume of distribution indicates extensive drug distribution into body tissues, suggesting the drug is primarily found in tissues rather than in the blood. Conversely, a low volume of distribution implies the drug remains mostly within the bloodstream.
Evaluating the volume of distribution offers several key advantages:
IMPACT-D is grounded in the world’s largest knowledge base on volume of distribution, known as PACT-D (Preclinical And Clinical Trials Knowledge Base on Volume of Distribution).
Reach out via our [contact form] for more details. We’d love to discuss the technology and explore collaboration opportunities with you.
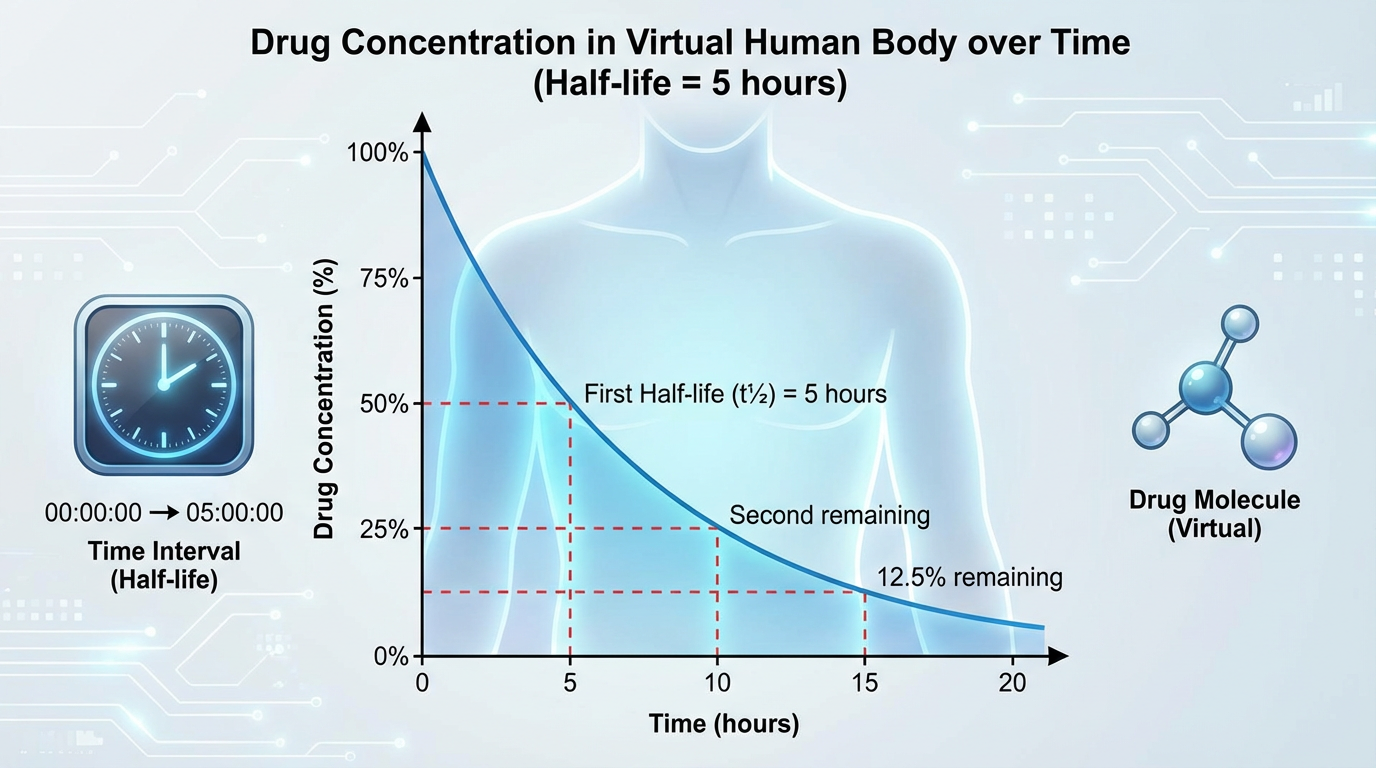
The expert system IMPACT-E predicts the terminal elimination half-life T1/2 of drug candidates in humans and animals, including monkeys, dogs, and rats.
The elimination half-life is the time required for the concentration of a drug in the blood to decrease by half. This is a crucial pharmacokinetic parameter for determining dosing schedules, estimating the duration of drug effects, and managing potential drug interactions.
IMPACT-E was developed from PACT-E, the world’s largest knowledge base on Elimination Half-life. Like all pharmacokinetic knowledge databases from PharmaInformatic, PACT-E is highly annotated, integrating all relevant information and conditions from preclinical and clinical studies available in the literature.
PharmaInformatic’s future expert systems can be tailored to meet specific customer requirements, such as oral versus IV drug administration, co-administration of drugs, gender differences, age, ethnicity, and more. Our structure-based knowledge bases contain comprehensive, annotated pharmacokinetic (PK) data.
Contact us through our [contact form] to learn more and explore potential collaborations.
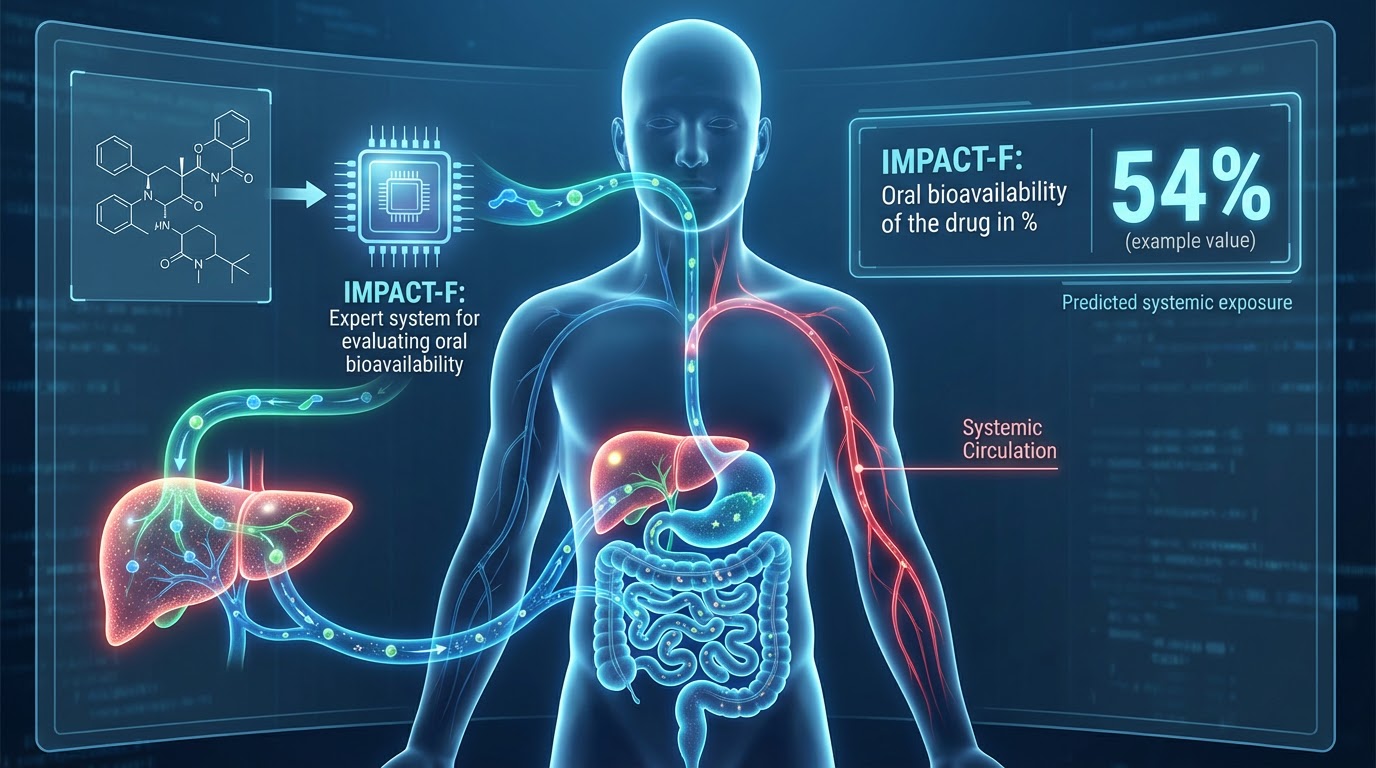
The expert system IMPACT-F assesses the oral bioavailability F% —the uptake of the parent, unchanged drug into the bloodstream—of a drug candidate in both humans and animals, including monkeys, dogs, rats, and mice. It is based on reliable computational models derived from the world’s largest knowledge base on bioavailability, known as PACT-F (Preclinical And Clinical Trials Knowledge Base on Bioavailability F%).
The early prediction of human oral bioavailability offers several advantages:
IMPACT-F has been employed by pharmaceutical companies in diverse therapeutic areas, including diabetes, inflammation, antivirals, autoimmune diseases, and cancer. It assists in selecting and prioritizing drug candidates, optimizing prodrugs, and evaluating oral bioavailability prior to human clinical trials. For more information, please refer to our collaborations or contact us via our [contact form]. We would be happy to present the technology and discuss potential collaborations at your convenience.
Link at collaborations to Collaborations
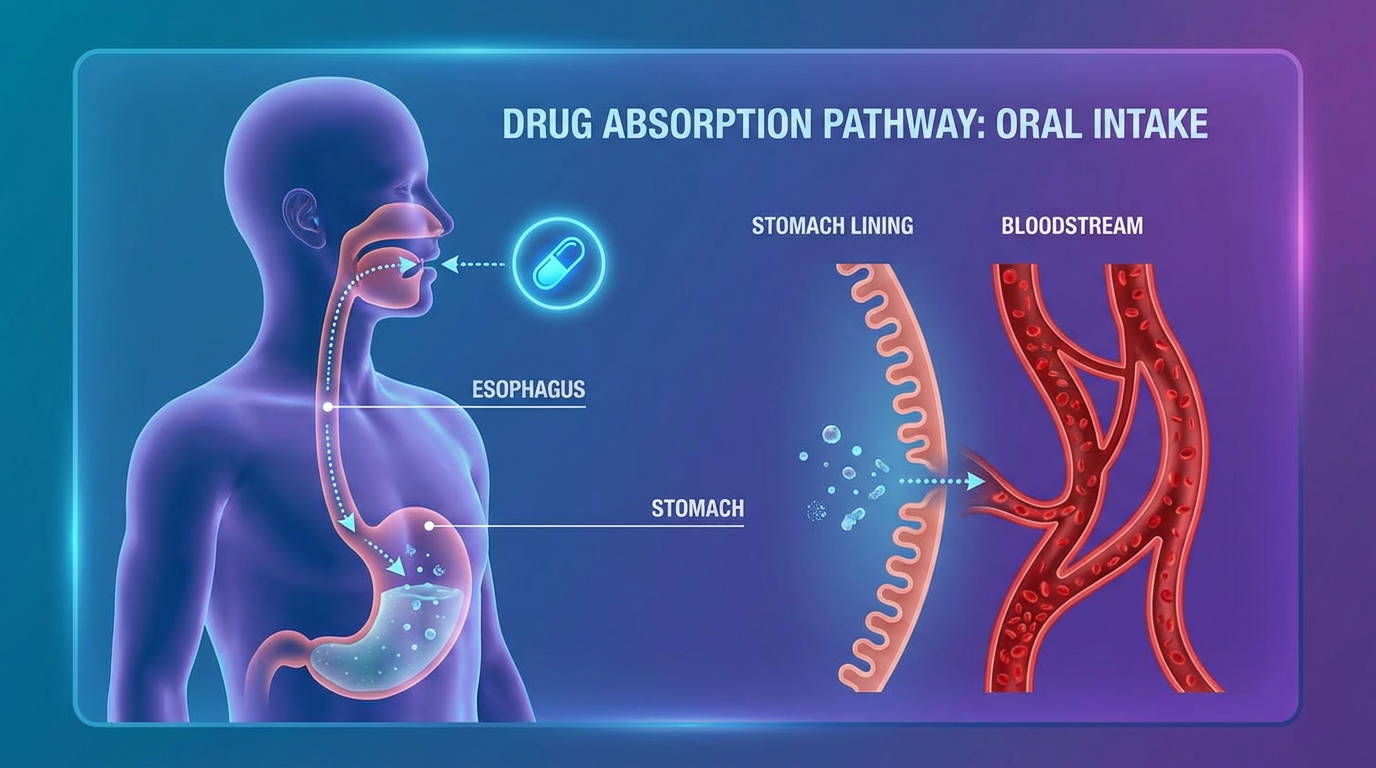
The expert system IMPACT-A (based on the knowledge base PACT-A) evaluates the Absorption of a small-molecule drug candidate into the human body. Oral drug absorption affects oral bioavailability, a key pharmacokinetic property of drugs. If a drug is not absorbed into the systemic circulation, it will have no systemic biological activity and will be excreted without producing any clinical effect.
The expert system IMPACT-F assesses the oral bioavailability—the uptake of the parent, unchanged drug into the bloodstream—of a drug candidate in both humans and animals, including monkeys, dogs, rats, and mice.
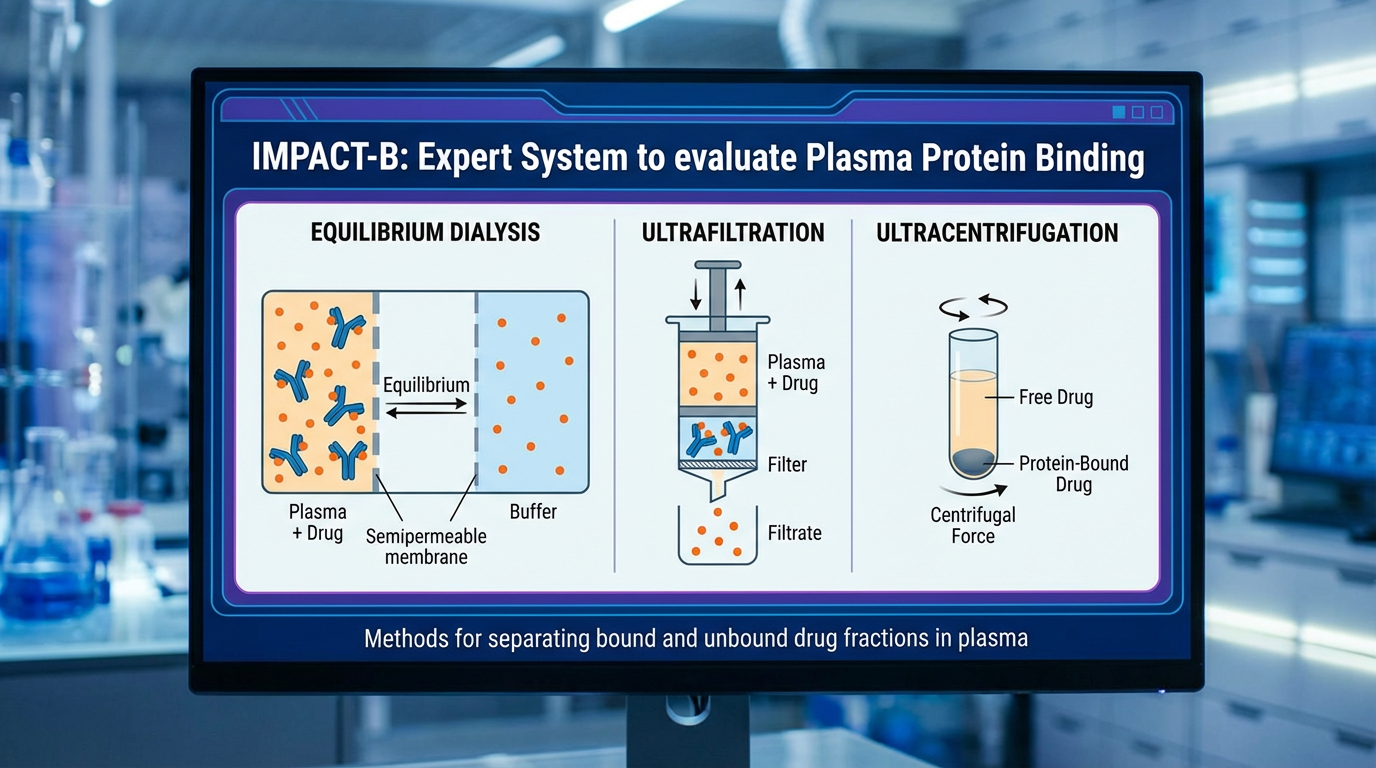
The expert system IMPACT-B evaluates the plasma protein binding PPB% of small molecules, such as chemical compounds or drugs, in humans.
In the bloodstream, drugs and toxic substances can bind to proteins, which decreases the free (active) concentration of these compounds in vivo. Understanding the extent to which a compound is bound to plasma proteins is crucial for toxicity evaluations and drug discovery because:
This AI-based technology was developed by PharmaInformatic as part of the German BMBF-funded EXITOX-II project.
Link to EXITOX-II project (see. Collaborations)
The AI-based expert system is grounded in PACT-B, the world’s largest knowledge base on Plasma Protein Binding.
IMPACT-B achieves outstanding predictive quality. Compared to conventional methods that estimate plasma protein binding based solely on compound structure, the error in predictions is reduced by half.
For further information, please contact us via our [contact form]. We would be happy to present the technology and discuss potential collaborations at your convenience.