Meet PharmaInformatic at BIO Europe Spring

Date: 23–25 March 2026 Location: Lisbon, Portugal Join us at BIO Europe Spring to explore how our cutting-edge solutions can accelerate your drug development projects. – Discuss collaboration opportunities tailored to your specific drug-candidate projects. – Evaluate any small molecule across a broad range of therapeutic areas with our advanced technologies. – Obtain clear and […]
Full-PK-KI-2: Replacing Animal Testing in Pharmacokinetics with Artificial Intelligence (2025-2027)
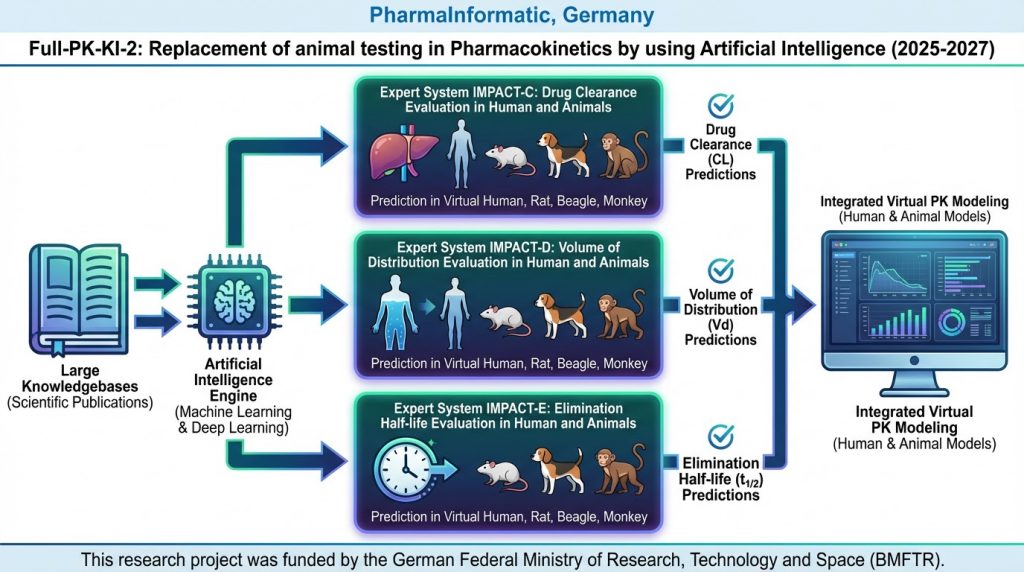
In the predecessor project, Full-PK-KI, expert systems were developed to accurately predict key pharmacokinetic parameters—distribution volume (VD), clearance (Cl), and elimination half-life (T1/2)—in humans, based exclusively on the structure of a drug candidate. Building on this foundation, the current project aims to further refine these expert systems by incorporating a broader array of drug structures […]
Full-PK-KI: Replacing Animal Testing in Pharmacokinetics with Artificial Intelligence (2022-2025)
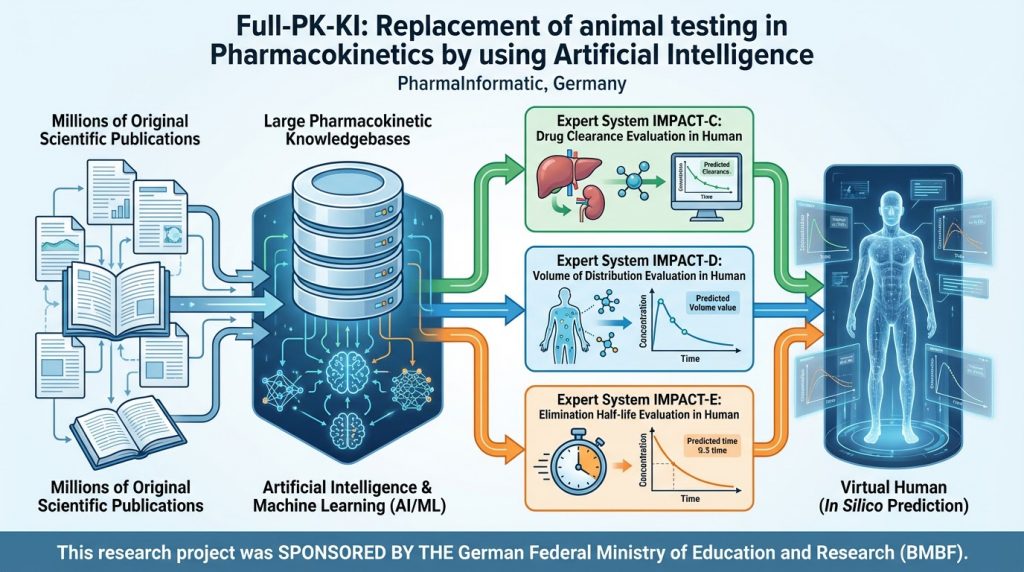
The drug uptake (bioavailability F), distribution (plasma protein binding PPB and volume of distribution VD), and elimination (clearance Cl and elimination half-life T1/2) are key pharmacokinetic properties essential for the development of new drugs. Traditionally, these properties are determined using animal studies in monkeys, dogs, rats, and mice to estimate pharmacological profiles in humans. PharmaInformatic […]
Herbert-Stiller Award 2021 to Wolfgang Boomgaarden from PharmaInformatic
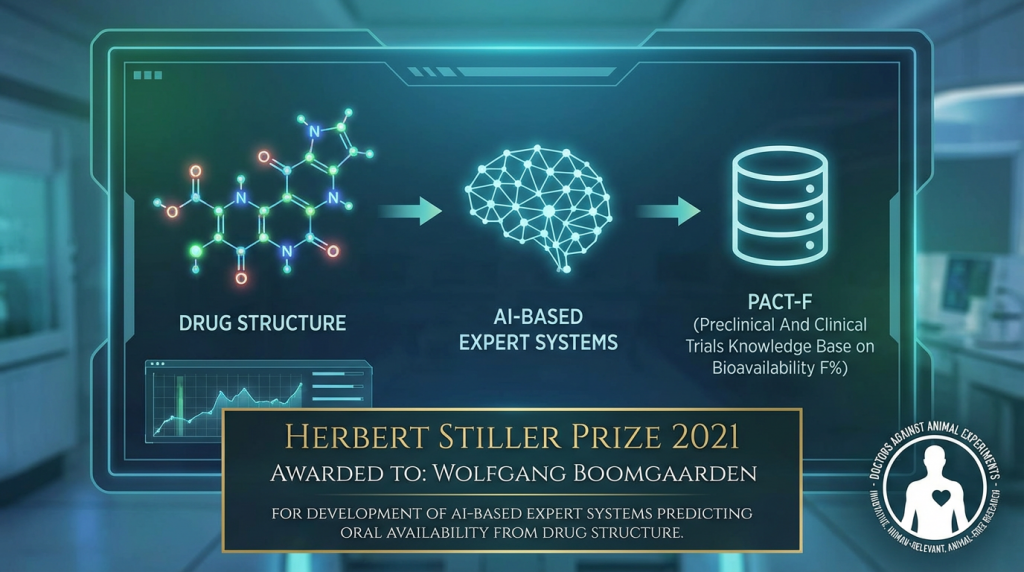
Good science requires good funding. This year, the nationwide association Doctors Against Animal Experiments is awarding two Herbert Stiller sponsorship prizes, each worth 20,000 Euros, for innovative, human-relevant, animal-free research. The winners have been announced: one award goes to MedUni Vienna, and the other is awarded to PharmaInformatic. Both projects have significant scientific potential and […]
