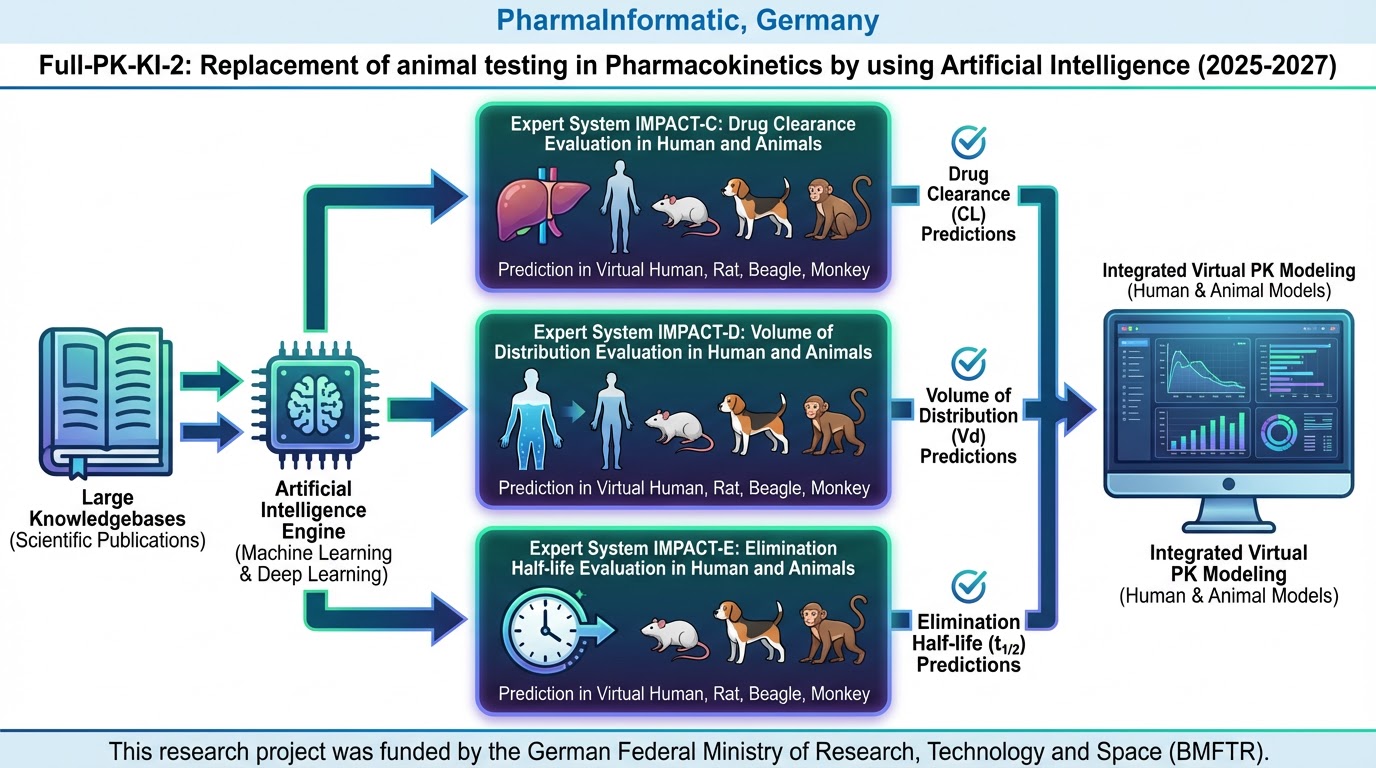
In the predecessor project, Full-PK-KI, expert systems were developed to accurately predict key pharmacokinetic parameters—distribution volume (VD), clearance (Cl), and elimination half-life (T1/2)—in humans, based exclusively on the structure of a drug candidate.
Building on this foundation, the current project aims to further refine these expert systems by incorporating a broader array of drug structures and their associated pharmacokinetic data. This enhancement will optimize both predictive accuracy and applicability.
Additionally, the research emphasized notable differences in pharmacokinetic properties between humans and various preclinical test animals. To address this, we are developing new expert systems to predict VD, Cl, and T1/2 specifically for rats, dogs, and monkeys.
This innovative approach enables pharmaceutical and biotech companies to conduct feasibility studies with the expert systems, validating them against their own existing preclinical drug results for these animals. This process can be handled in-house, ensuring that confidential data remains protected.
If validation results align closely with expectations, these expert systems can be seamlessly integrated into the planning of new clinical trials in humans, thus providing direct support and enhancing efficiency in the drug development process.

This research project was funded by the German Federal Ministry of Research, Technology and Space (BMFTR).
Reach out via our contact form for more details. We’d love to discuss the technology and explore collaboration opportunities with you.