- determines the effective concentration in human Cfree (unbound concentration)
- technology combines different AI-based approaches, but results are almost immediately available
- improves QIVIVE & toxicity evaluations, evaluates protein binding before a compound is needed to be synthesized and predicts protein binding of unstable or reactive compounds in human
Expert system
Protein Binding Prediction of compounds in human
Drugs and toxic substances can be bound to proteins in plasma or blood, which reduces the free (effective) concentration of compounds in vivo. For toxicity evaluations and in drug discovery it is important to know to which extend a compound is bound to plasma proteins (PPB%):

To use our prediction service please give us a call or send an email by clicking the button.
The expert system has been derived from the largest knowledge base on experimental protein binding data worldwide.
 The development was funded partly by the German Federal Ministry of Education and Research (BMBF) within the funding program e:ToP Innovative Toxikologie zur Reduzierung von Tierversuchen. An outstanding quality of prediction was achieved: Compared to conventional methods which forecast plasma protein binding by compound structure the error of prediction was reduced by half.
The development was funded partly by the German Federal Ministry of Education and Research (BMBF) within the funding program e:ToP Innovative Toxikologie zur Reduzierung von Tierversuchen. An outstanding quality of prediction was achieved: Compared to conventional methods which forecast plasma protein binding by compound structure the error of prediction was reduced by half.
The technology, originally developped to replace animal tests, now helps to identify effective antiviral drugs for the treatment of COVID-19. In combination with the expert system IMPACT-F the effectice concentration in human blood resulting from an oral dose can be estimated of any small-molecule drug candidate:
Clinical failures can be identified with AI-based expert systems BEFORE human clinical trials are carried out
New potent drug candidates against coronavirus must achieve sufficient effective concentration in blood, high enough to erase the virus in humans. The effective concentration in human blood is reduced by low drug uptake (oral bioavailability) and high plasma protein binding. Our AI-based technology can calculate these key PK properties for any small-molecule drug candidate in almost all therapeutic areas. A current example is given below: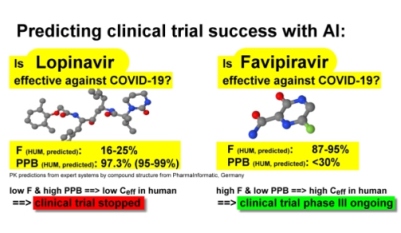 The pharmacokinetic evaluation of two current drug candidates for the treatment of COVID-19 with AI-based expert systems showed that the effective concentration in human blood of Lopinavir would be too low to be effective in humans due to low oral bioavailability F and high plasma protein binding PPB.
The pharmacokinetic evaluation of two current drug candidates for the treatment of COVID-19 with AI-based expert systems showed that the effective concentration in human blood of Lopinavir would be too low to be effective in humans due to low oral bioavailability F and high plasma protein binding PPB.
On the other hand, the predicted oral bioavailability F of Favipiravir was high and predicted plasma protein binding PPB was low leading to a much higher effective concentration in humans.
If the effective concentration in human blood is too low, the drug cannot work and human clinical trials will be not successful.
Results of current clinical trials:
Clinical trials with Lopinavir were stopped due to lack of efficacy against COVID-19 (details).
A clinical trial phase III was started with Favipiravir against COVID-19 in April 2021 (details).


|
© Copyright 2004-2021 PharmaInformatic Boomgaarden. All rights reserved. Site map Contact Terms of Use Imprint |
 |
|
|
|
|
QIVIVE
Quantitative In Vitro to In Vivo Extrapolation
In order to correlate
IN VITRO results
with
IN VIVO observations, plasma protein binding
of a compound
is needed.
The effective
concentration
Cfree or Cunbound
can be largely different
from the nominal concentration
of compounds due to
plasma protein binding.
