|
|
|
|
Lead optimization and drug-candidate selection |
|
|
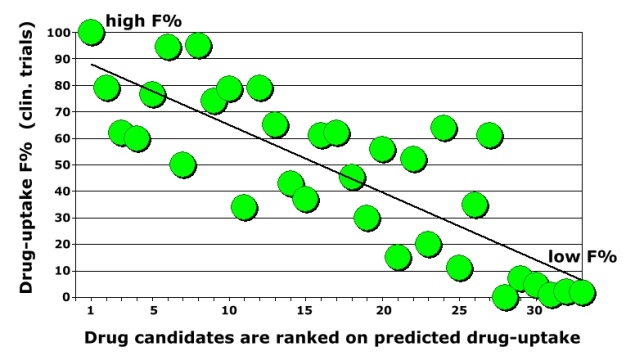
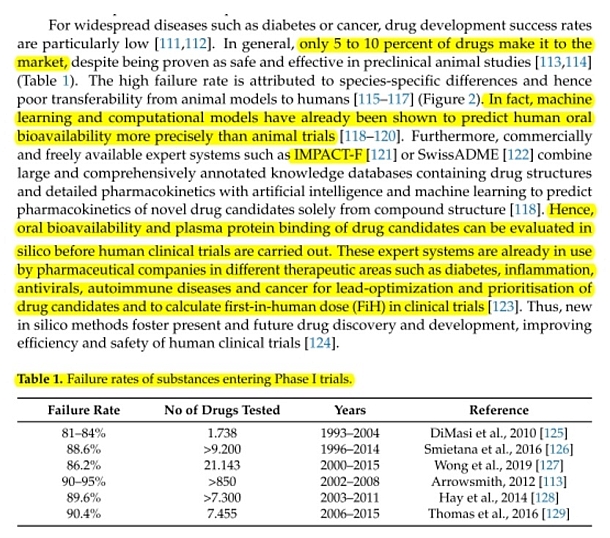
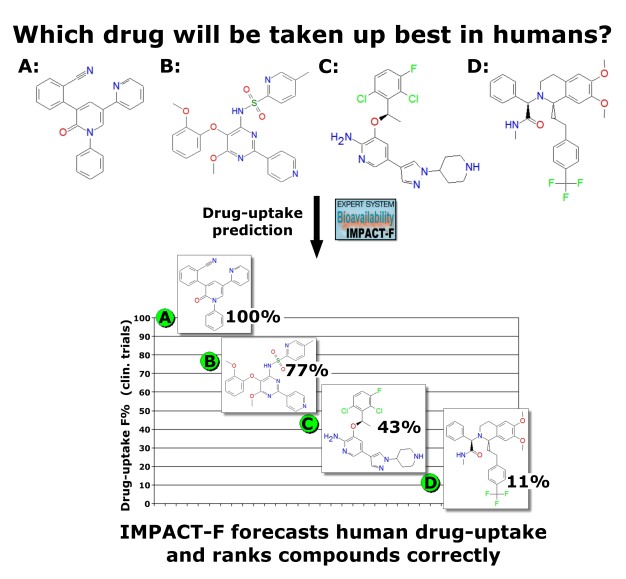
A drug must have sufficient oral bioavailability in humans otherwise clinical trials are not effective and drug development is then stopped. Oral bioavailability is one of the most important properties in lead optimisation. Low drug-uptake in humans can result in high inter-individual variability and increases the risk of side-effects and toxicity. IMPACT-F correctly ranks (prioritises) novel drug-candidates on human oral bioavailability. A validation study was carried out containing thirty-three independent drug-candidates coming from different therapeutic areas such as cancer, antibacterial or antiviral infection, CNS, inflammation, gastrointestinal and renal diseases, metabolism related diseases and cardiovascular diseases. |
|
|
|
|
|
A low error and a good correlation was observed. Oral bioavailability predictions were as accurate as the common deviation between individual humans in a clinical trial. IMPACT-F ranks compounds with global QSPR models so only drug structures are needed to carry out a prioritisation study for selecting leads and drug-candidates. |
|
|
To use our prediction service or to obtain more information, please give us a call (+49 4921-993360) or send an email by clicking the button: |
|
|
|
|
|
|
|
|
The expert system reliably evaluates drug-candidates ranging across all therapeutic areas. The validation set included a CCR5 antagonist for treatment of rheumatoid arthritis, a p38 MAP-kinase inhibitor for treatment of acute coronary syndrome, four novel angiotensin II receptor blocker (antihypertensive), a beta3-adrenoceptor agonist in development for treatment of overactive bladder, two novel HIV-1 non-nucleoside reverse transcriptase inhibitors (antivirals), a dual orexin receptor antagonist, three different cardiovascular drugs (treatment of atrial fibrillation, neutrophil elastase inhibitors), three new cancer drugs (an ALK and ROS1 inhibitor for the treatment of carcinoma, an ERK kinase 1 and 2 inhibitor and a BRAF inhibitor), an endothelin A receptor blocker, two novel factor Xa inhibitors (anticoagulants), a motilin receptor agonist (gastrointestinal), a marketed thyroid hormone, an antiepileptic agent (AMPA antagonist), two novel dipeptidyl-peptidase-4-inhibitors for treatment of type 2 diabetes, a PDE4 inhibitor for treatment of COPD and asthma, a JAK3 inhibitor to treat rheumatoid arthritis, a histamine H1 antagonist, a microsomal triglyceride transfer protein inhibitor, a traded vitamin, a drug to treat electrolyte metabolic disorders, an anticonvulsant clinical candidate (potassium agonist) and two antibacterial drugs (a traded drug inhibiting DNA-dependent RNA polymerase and an investigational peptide deformylase inhibitor). |
|
|
|
|
|
Current Review article on oral bioavailability in Pharmaceutics 2021: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
IMPACT-F optimizes lead candidates in drug discovery |
|
|
|
|
|
The artificial intelligence technology, developed by PharmaInformatic, Germany, evaluates if a potential drug will be efficiently taken up in humans. IMPACT-F prioritizes drug-candidates based on predicted drug-uptake (oral bioavailability). This ranking allows the most efficient drug-candidates to be chosen for further development. |
|
|
|
|
|
|
|
|
The technology has been used by pharmaceutical companies in therapeutic areas such as diabetes, inflammation, autoimmune diseases, antivirals and cancer. It has been applied to optimize lead candidates and to evaluate oral bioavailability and effective dose prior to human clinical trials. The first drug candidates evaluated with IMPACT-F have now progressed into clinical trials in humans. |
Current Press Release:
IMPACT- F optimizes
lead candidates
in drug discovery
(pdf)![]()
LEAD
OPTIMIZATION
with
IMPACT-F:
==> selects oral bioavailable
drugs
==> increases efficacy of clinical trials
==> results
directly available
Only compound structure needed
==> no further assays, synthesis
or costs
Explanations
&
Abbreviations
ADME
Absorption Distribution Metabolism Excretion.
Oral
Bioavailability combines Absorption & Metabolism
PK Pharmacokinetics
QSAR
Quantitative Structure
Activity
Relationship
The chemical structure is set in relation to
biological activity.
IND
Investigational
New Drug
Lead Optimization
Drug discovery starts from
initial hits with pharmacological activity.
Structural modifications of compound structure yield to optimised leads. They can progress to drug candidates, which are investigated in human clinical trials.
DMPK
Drug Metabolism and Pharmacokinetics
NDA
New Drug Application
QSPR
Quantitative Structure
Property Relationship
Oral
Bioavailability
is key property
of leads and
drug-candidates.
NCE
New Chemical Entity
Novel models to forecast
Plasma Protein Binding
of compounds
show a high quality of prediction.
A low error
and high
correlation
between experimental and predicted PPB values was found, based on a large independent validation dataset.


|
© Copyright 2004-2021 PharmaInformatic Boomgaarden. All rights reserved. Site map Contact Terms of Use Imprint |
 |
|
|
|
|
|
|
Bioavailability prediction of drug candidates for
treatment of Type 2 diabetes
(more)
...and in short time our development project was taken to the next step."
New collaboration:
CRACK IT
DoCE Challenge
(Unilever, Shell)
Sponsored by Unilever and Shell, this challenge aims to develop methods and tools that will better quantify and control the exposure of chemicals for predicting in vivo toxicity.