- selection of bioavailable drug candidates ==> reduces clinical failures, more reliable results than animal experiments, results are almost immediately available
- increases the prospects of novel drugs, because the optimum oral dose for first-in-human clinical trials can be determined much more accurately.
- unrivalled method to evaluate efficiency of drugs before clinical trials in humans, confidential and reliable (no data or information leaves your company)
IMPACT-F
Predicting human oral bioavailability of future drug candidates
The evaluation of oral bioavailability is based on reliable computational models, which have been derived from the largest knowledge base on bioavailability worldwide (PACT-F). The early prediction of human oral bioavailability has several advantages:
IMPACT-F is used by pharmaceutical companies in different therapeutic areas such as diabetes, inflammation, antivirals, autoimmune diseases and cancer for selection and prioritisation of drug candidates, to optimise prodrugs and to evaluate oral bioavailability before clinical trials in humans, see recent collaborations.
To use our prediction service or to obtain more information, please give us a call (+49 4921-993360) or send an email by clicking the button:

IMPACT-F:
Download
product flyer
(pdf)
Prediction of oral bioavailability reduces failures in clinical trials.
Related Links:
Cheminformatics
ADME-Software
Pharmacokinetics
Computational chemistry
Molecular
modeling
IND:
Investigational new drug
NCEs:
New chemical entities
DMPK:
Drug
metabolism
and pharmacokinetics
NDA:
New drug
application
Oral bioavailability is a key property in drug design and development. Poor bioavailability of a drug results in low efficacy and higher inter-subject variability and therefore can lead to unpredictable response to a drug.
The expert system IMPACT-F is easy to use, requires no chemical synthesis or animal experiments, is much more reliable than animal trials and results are almost immediately available. It is an important method in future drug discovery and development which improves efficiency and safety of human clinical trials.
Current Review article on oral bioavailability and regulatory aspects published in Pharmaceutics 2021:
For widespread diseases such as diabetes or cancer, drug development success rates are particularly low [111,112]. In general, only 5 to 10 percent of drugs make it to the market, despite being proven as safe and effective in preclinical animal studies [113,114] (Table 1). The high failure rate is attributed to species-specific differences and hence poor transferability from animal models to humans [115117] (Figure 2).
In fact, machine learning and computational models have already been shown to predict human oral bioavailability more precisely than animal trials [118120]. Furthermore, commercially and freely available expert systems such as IMPACT-F [121] or SwissADME [122] combine large and comprehensively annotated knowledge databases containing drug structures and detailed pharmacokinetics with artificial intelligence and machine learning to predict pharmacokinetics of novel drug candidates solely from compound structure [118].
Hence, oral bioavailability and plasma protein binding of drug candidates can be evaluated in silico before human clinical trials are carried out. These expert systems are already in use by pharmaceutical companies in different therapeutic areas such as diabetes, inflammation, antivirals, autoimmune diseases and cancer for lead-optimization and prioritisation of drug candidates and to calculate first-in-human dose (FiH) in clinical trials [123]. Thus, new in silico methods foster present and future drug discovery and development, improving efficiency and safety of human clinical trials [124].
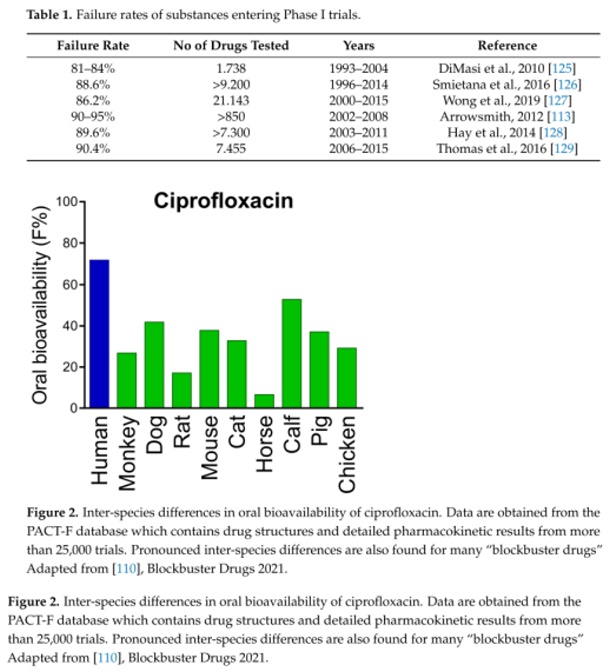
Reference: Zietek T, Boomgaarden WAD, Rath E. Drug Screening, Oral Bioavailability and Regulatory Aspects: A Need for Human Organoids. Pharmaceutics. 2021; 13(8):1280. https://doi.org/10.3390/pharmaceutics13081280
|
Frequently Asked Questions (FAQ):
|
|
Can IMPACT-F predict human oral bioavailability of entirely new molecules? |
|
Is IMPACT-F restricted to a specific therapeutic area, such as antivirals or cancer? |
|
What is required to predict oral bioavailability with IMPACT-F? |
|
At which stage of drug discovery and development should IMPACT-F be applied? |
|
|
|
Question: My company has discovered a new chemical entity (NCE) with a novel and unique mechanism of action. Up to now, no similar molecules have been investigated in human clinical trials. Can IMPACT-F predict human oral bioavailability of entirely new molecules? Answer: Yes. In cooperation with a pharmaceutical company, we have investigated this question. We have found that constantly good prediction results are obtained, regardless of whether highly similar compounds are present in PACT-F, the underlying knowledge base of IMPACT-F. In fact, more than 82% of the investigated drug-candidates showed a maximal Tanimoto similarity below 0.67. This shows that highly similar drugs are not needed to reliably estimate the oral bioavailability of entirely novel drug structures. Please contact us if you are interested in the detailed report. On the other hand, we found that small changes in molecular structure can dramatically influence the oral bioavailability of a drug candidate. IMPACT-F uses and combines all available information in PACT-F from previous known bioavailability to create a reliable estimate of oral bioavailability in humans.
|
|
Question: Is IMPACT-F restricted to a specific therapeutic area, such as antibiotics, cardiovascular drugs, or cancer therapeutics? Answer: No, there is no restriction on a specific therapeutic area. IMPACT-F can predict the oral bioavailability of small-molecule drug-candidates ranging across all therapeutic areas. We have evaluated IMPACT-F with drugs and drug-candidates from all therapeutic areas, such as cardiovascular, cancer, gastrointestinal, pain, antivirals, antibacterials, inflammation, anticoagulants, antiepileptic drugs, Alzheimers and Parkinsons disease, renal diseases, Multiple Sclerosis, respiratory diseases, antidiabetic agents, COPD, asthma, rheumatoid arthritis, obesity, metabolic disorders, central nervous system disorders. Reliable prediction results were achieved in all cases, so there is no restriction to a specific therapeutic area. IMPACT-F has been used by pharmaceutical companies in different therapeutic areas (cancer, diabetes, inflammation, autoimmune diseases, antivirals) for selection and prioritisation of drug candidates, to optimise prodrugs and to evaluate oral bioavailability before advanced clinical trials in humans, see recent collaborations.
|
|
Question: What is required to predict oral bioavailability with IMPACT-F (in vivo PK in rats, measured logP)? Answer: The structure of the drug-candidate and time to do the calculations, thats all. Eight years ago, our aim was to create an expert system which was not dependent on additional experimental input parameters. They take time to generate, different conditions in measurement cannot always be unified, and chemical synthesis is expensive and takes time as well. The chemical structure of an investigational compound is available at a very early time in drug discovery projects, even before chemical synthesis and preclinical experiments can take place. If the decision to develop a compound further can be made at the earliest possibility, then more money and time can be saved and resources can be concentrated on the most prospective ones. In drug discovery it is important to identify useful drug candidates at the earliest opportunity. A large amount of time and money is spent on candidates which will never come to market. Low oral bioavailability in clinical trials is one of the main reasons why clinical trials fail.
|
|
Question: At which stage of drug discovery and development should IMPACT-F be applied? Answer: The earlier the better! Many drug-candidates fail in clinical trials due to low oral bioavailability in humans. As a result billions of Euros are wasted every year on drug-candidates which never reach the market. IMPACT-F identifies these failures and substantially reduces drug development costs and time involved. IMPACT-F has now been used by several pharmaceutical companies, even though the technology was launched only a few months ago. Up to now, the expert system was applied in different phases of drug discovery and development. On the one hand within the preclinical discovery phase to optimise and select drug candidates with high oral bioavailability. On the other hand IMPACT-F has estimated the oral bioavailability of drug-candidates which are scheduled for clinical trials in humans.
Further explanations and abbreviations: |
IMPACT-F:
Download
product flyer
(pdf)
Prediction of oral bioavailability reduces clinical failures and increases safety and prospects of clinical trials.
Frequently Asked Questions (FAQ)
New collaboration:
CRACK IT
DoCE
Challenge
Sponsored by Unilever
& Shell
this challenge aims to develop methods and tools that will better quantify and control the exposure of chemicals for predicting in vivo toxicity.


|
© Copyright 2004-2021 PharmaInformatic Boomgaarden. All rights reserved. Site map Contact Terms of Use Imprint |
 |
|
|
|
|
|
|
Bioavailability prediction of drug candidates for
treatment of Type 2 diabetes
(more)
...and in short time our development project was taken to the next step."
 |
|
|
|
|
Recent News:
Selection and prioritisation of lead compounds for the treatment of inflammatory and autoimmune diseases based on estimated oral bioavailability in humans with IMPACT-F.
(more)
Novel models to forecast
Plasma Protein Binding
of compounds
show a high quality of prediction.
Based on a large
independent validation dataset.
a low error
and high correlation
between experimental and predicted PPB values was found.
 |
Selection and prioritisation
of prodrugs
for the treatment of
cancer
based on estimated oral bioavailability in humans
with IMPACT-F.
(more)

